issue contents
January 2018 issue
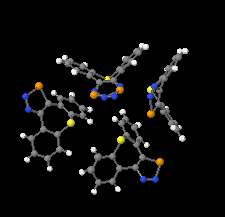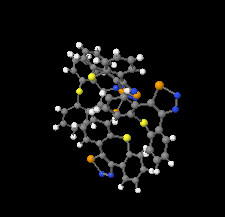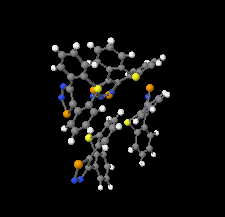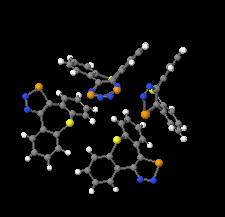
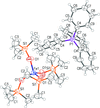
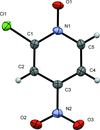
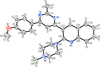
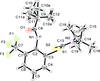
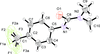
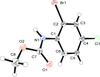
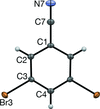
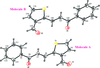
Cover illustration: 1,2,3-Selenadiazoles are useful precursors for strained cycloalkynes and heterocycles, and are precursors for photochemically induced as well as for strain-accelerated click-reactions. Sixteen molecules, i.e. four sets of four independent molecules, of dibenzo[2,3:6,7]thiepino[4,5-d][1,2,3]selenadiazole fill the unit cell (Z' = 4). The four independent tetracyclic molecules have very similar molecular structures, each adopting a saddle shape. In the crystal, layers of molecules with hydrogen surfaces extend parallel to the ab plane and are stacked along the c axis. See: Schollmeyer & Detert [IUCrData (2017). 3, x180070].
inorganic compounds


 access
accessmetal-organic compounds


 access
access

 access
access

 access
access

 access
access

 access
access

 access
accessorganic compounds


 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 journal menu
journal menu












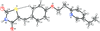




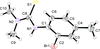
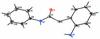

![[publCIF]](/logos/authorchecklist11.gif)