issue contents
December 2017 issue
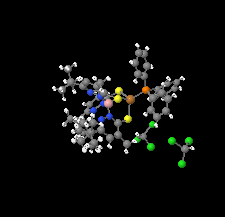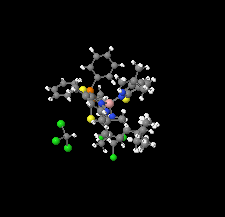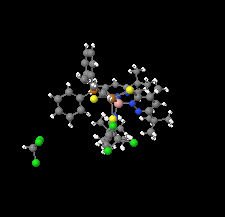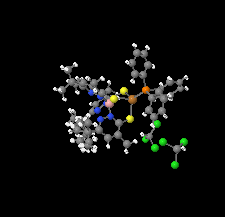
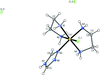
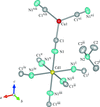
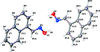
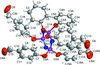
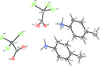
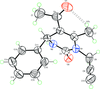
Cover illustration: Soft scorpionate ligands have gained significant attention in synthetic inorganic chemistry over the last decades. The reaction of the soft scorpionate ligand potassium tris(6-tert-butyl-4-methylpyridazine-3(2H)-thione)borate (= KTnMe,tBu) with copper(I) chloride in the presence of triphenylphosphane retained the borate ligand and led to the compound {(hydrogen 2,2',2''-boranetriyl)tris[6-tert-butyl-4-methylpyridazine-3(2H)-thione]-![[kappa]](/logos/entities/kappa_rmgif.gif) 3H,S,S'}(triphenylphosphane-
3H,S,S'}(triphenylphosphane-![[kappa]](/logos/entities/kappa_rmgif.gif) P)copper(I) chloroform disolvate; this represents an illustrative example where the scorpionate ligand binds in a
P)copper(I) chloroform disolvate; this represents an illustrative example where the scorpionate ligand binds in a ![[kappa]](/logos/entities/kappa_rmgif.gif) 3-H,S,S fashion, in contrast to previously reported copper complexes with this ligand where boratranes (Cu-B bond) were found exclusively. The CuI atom is surrounded in a trigonal-planar arrangement by the triphenylphosphane ligand and two of the three S atoms of the scorpionate ligand with an additional 3-center-2-electron B-H
3-H,S,S fashion, in contrast to previously reported copper complexes with this ligand where boratranes (Cu-B bond) were found exclusively. The CuI atom is surrounded in a trigonal-planar arrangement by the triphenylphosphane ligand and two of the three S atoms of the scorpionate ligand with an additional 3-center-2-electron B-H Cu interaction, with the H atom as the apex of a pyramid. See: Holler, Belaj & Mösch-Zanetti [IUCrData (2017). 2, x171772].
Cu interaction, with the H atom as the apex of a pyramid. See: Holler, Belaj & Mösch-Zanetti [IUCrData (2017). 2, x171772].
inorganic compounds


 access
accessmetal-organic compounds


 access
access

 access
access

 access
access

 access
access

 access
accessorganic compounds


 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 journal menu
journal menu











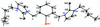


![[publCIF]](/logos/authorchecklist11.gif)