issue contents
September 2017 issue
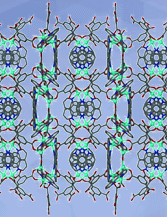
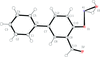
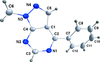
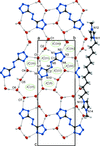
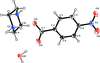
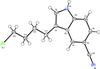
Cover illustration: 4-Cyano-3,5-bis(phenyl)pyrazole, a symmetrically phenyl substituted 4-cyano pyrazole, was prepared as a precursor for the preparation of scorpionate ligands. The C-C![[triple bond]](/logos/entities/z-tbnd_rmgif.gif) N fragments in the two independent molecules in the asymmetric unit deviate only slightly from linearity, with bonding angles of 179.4 (2) and 178.9 (2)°. In the crystal, molecules are linked by pairs of N-H
N fragments in the two independent molecules in the asymmetric unit deviate only slightly from linearity, with bonding angles of 179.4 (2) and 178.9 (2)°. In the crystal, molecules are linked by pairs of N-H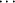 N hydrogen bonds, forming inversion dimers with an R22(6) ring motif. See: Zhao, Adeyemi & Pompilius [IUCrData (2017). 2, x171257].
N hydrogen bonds, forming inversion dimers with an R22(6) ring motif. See: Zhao, Adeyemi & Pompilius [IUCrData (2017). 2, x171257].
metal-organic compounds


 access
access

 access
access

 access
access

 access
access

 access
accessorganic compounds


 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
accessaddenda and errata


 access
access

 journal menu
journal menu











![[publCIF]](/logos/authorchecklist11.gif)