issue contents
September 2019 issue
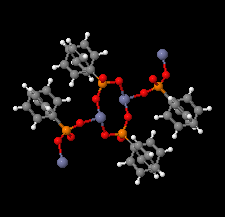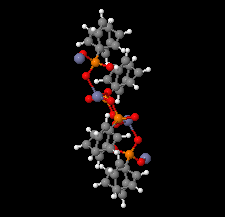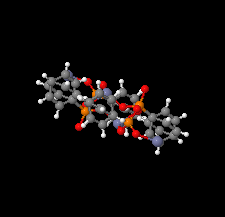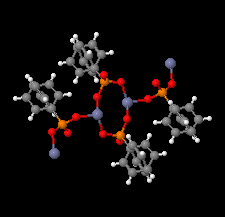
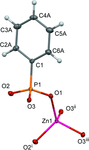
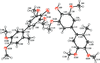
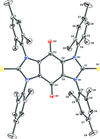
Cover illustration: The coordination polymer poly[(![[mu]](/logos/entities/mu_rmgif.gif) 4-phenylphosphonato)zinc(II)], [Zn(C6H5O3P)], was obtained serendipitously by reacting zinc(II) acetate with a tetraphosphonate cavitand in DMF. The basic conditions of the reaction cleaved the phosphonate bridges at the upper rim of the cavitand, making them available for reaction with the zinc ions. The coordination polymer can be described as an inorganic layer in which zinc coordinates the oxygen atoms of the phosphonate groups in a distorted tetrahedral environment, while the phenyl groups, which are statistically disordered over two orientations, point up and down with respect to the layer. The layers interact through van der Waals interactions. Data analysis, structure solution, refinement and validation were conducted as part of a tutorial session during the Crystallographic Information Fiesta held in Naples, Italy, from 29 August to 3 September 2019, and organized by the Italian Crystallographic Association in partnership with the IUCr. See: Falvello, Lotti, Massera, Tarantino, Zema, Puschmann, Agbahoungbata, Andreo, Ashoka Sahadevan, Bismuto, Bonfant, Bonou, Carraro, De Zotti, di Biase, Fantini, Ferraboschi, Ferreira Custodio, Frigerio, Gallo, Gjyli, Goudjil, Igoa, Kahveci, Kalienko, Lorenzon, Macera, Manrique Fajardo, Nushi, Ouaatta, Parisi, Pasqualetto, Pesko, Pierri, Pinalli, Poppe, Santoro, Smirnova, Sorbara, Tensi & Tusha [IUCrData (2019). 4, x191222].
4-phenylphosphonato)zinc(II)], [Zn(C6H5O3P)], was obtained serendipitously by reacting zinc(II) acetate with a tetraphosphonate cavitand in DMF. The basic conditions of the reaction cleaved the phosphonate bridges at the upper rim of the cavitand, making them available for reaction with the zinc ions. The coordination polymer can be described as an inorganic layer in which zinc coordinates the oxygen atoms of the phosphonate groups in a distorted tetrahedral environment, while the phenyl groups, which are statistically disordered over two orientations, point up and down with respect to the layer. The layers interact through van der Waals interactions. Data analysis, structure solution, refinement and validation were conducted as part of a tutorial session during the Crystallographic Information Fiesta held in Naples, Italy, from 29 August to 3 September 2019, and organized by the Italian Crystallographic Association in partnership with the IUCr. See: Falvello, Lotti, Massera, Tarantino, Zema, Puschmann, Agbahoungbata, Andreo, Ashoka Sahadevan, Bismuto, Bonfant, Bonou, Carraro, De Zotti, di Biase, Fantini, Ferraboschi, Ferreira Custodio, Frigerio, Gallo, Gjyli, Goudjil, Igoa, Kahveci, Kalienko, Lorenzon, Macera, Manrique Fajardo, Nushi, Ouaatta, Parisi, Pasqualetto, Pesko, Pierri, Pinalli, Poppe, Santoro, Smirnova, Sorbara, Tensi & Tusha [IUCrData (2019). 4, x191222].
metal-organic compounds
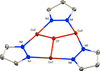
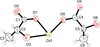
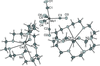
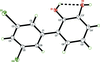
The centrosymmetric molecule of the title compound can be regarded as a dimer, consisting of two pairs of copper(II) cations arranged in a triangle and bridged by pyrazolate, carboxylate groups and hydroxido ligands.
L. Falvello,
P. Lotti,
C. Massera,
S. C. Tarantino,
M. Zema,
H. Puschmann,
M. Y. Agbahoungbata,
J. Andreo,
S. A. Sahadevan,
A. Bismuto,
G. Bonfant,
S. A. S. Bonou,
C. Carraro,
M. D. Zotti,
A. di Biase,
R. Fantini,
I. Ferraboschi,
J. M. F. Custodio,
M. Frigerio,
G. Gallo,
S. Gjyli,
M. Goudjil,
F. Igoa,
E. Kahveci,
M. Kalienko,
S. Lorenzon,
L. Macera,
J. J. M. Fajardo,
E. Nushi,
S. Ouaatta,
E. Parisi,
L. Pasqualetto,
E. Pesko,
G. Pierri,
R. Pinalli,
R. Poppe,
A. Santoro,
E. Smirnova,
S. Sorbara,
L. Tensi and
G. Tusha
The title coordination polymer was obtained reacting zinc(II) acetate with a tetraphosphonate cavitand in DMF. The basic conditions of the reaction medium hydrolysed the cavitand making the phosphonate group available for coordination to the zinc(II) cation.
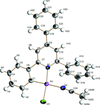
The structure of a salt consisting of a tetraisothiocyanatocobalt(II) complex anion, a water-bonded postassium cation and a D-(+)-glucosammonium cation was refined by single-crystal X-ray diffraction analysis in the P212121 space group.
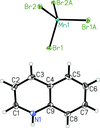
The title compound consists of two quinolinium cations and an MnBr4 anion, which exhibits a distorted tetrahedral coordination geometry.
The cadmium cations (coordination number seven) are linked by hydroxypropanoate anions into a three-dimensional network.
The pentacarbonyl molybdate complex [Cs([2.2.2]crypt)]2[Mo(CO)5] was obtained by the reduction of Mo(CO)3(η5-Cp)H with Cs5Bi4 in the presence of [2.2.2]crypt in liquid ammonia and contains an approximate trigonal–bipyramidal [Mo(CO)5]2− anion.
In the crystal, molecules are arranged via a metallophilic interaction between platinum atoms with a Pt⋯Pt contact of 7.052 (2) Å. In addition, a π–π interaction occurs.
organic compounds
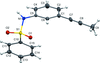
The synthesis of the title sulfanilamide derivative and the determination of its crystal structure are reported.
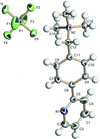
In the cation of the title molecular salt, C15H19N2+·PF6−, the dihedral angle between the benzene and pyridine rings is 38.21 (10)°.
The asymmetric unit of the title compound contains two independent molecules,
A and
B, with similar geometries. In the crystal, the
A molecules form [1
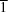
0] chains linked by O—H⋯O hydrogen bonds from the hydroxyl group to one of the methoxy O atoms. The
B molecules form O—H⋯O hydrogen bonds to the hydroxyl O atoms of the
A molecules and thus act as fixed spacers between the chains of molecule
A.
The title structure, a putative metabolite of 3,5-dichlorobiphenyl (PCB 14), the dihedral angle between the two benzene rings of the title compounds is 58.86 (4)°.
Very weak C—H⋯N and C—H⋯O interactions occur in the crystal structure of the title compound.
Crystallization from a commercial source of 3-ethyl-4-methyl-3-pyrrolin-2-one afforded 1:1 co-crystals of this compound (C7H11NO) with its oxidized derivative, 3-ethyl-4-methyl-3-pyrrolin-2,5-dione (C7H9NO2).
The title compound, 1,1′,3,3′-tetramesitylquinobis(imidazole)-2,2′-dithione [+solvent], crystallizes in the cubic space group
Im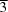
. The crystal structure exhibits large cavities of 193 Å
3 accounting for 7.3% of the total unit-cell volume.
The synthesis, molecular and crystal structure of a biologically important pharmacophore containing chalcone is reported

![[mu]](/logos/entities/mu_rmgif.gif) 4-phenylphosphonato)zinc(II)], [Zn(C6H5O3P)], was obtained serendipitously by reacting zinc(II) acetate with a tetraphosphonate cavitand in DMF. The basic conditions of the reaction cleaved the phosphonate bridges at the upper rim of the cavitand, making them available for reaction with the zinc ions. The coordination polymer can be described as an inorganic layer in which zinc coordinates the oxygen atoms of the phosphonate groups in a distorted tetrahedral environment, while the phenyl groups, which are statistically disordered over two orientations, point up and down with respect to the layer. The layers interact through van der Waals interactions. Data analysis, structure solution, refinement and validation were conducted as part of a tutorial session during the Crystallographic Information Fiesta held in Naples, Italy, from 29 August to 3 September 2019, and organized by the Italian Crystallographic Association in partnership with the IUCr. See: Falvello, Lotti, Massera, Tarantino, Zema, Puschmann, Agbahoungbata, Andreo, Ashoka Sahadevan, Bismuto, Bonfant, Bonou, Carraro, De Zotti, di Biase, Fantini, Ferraboschi, Ferreira Custodio, Frigerio, Gallo, Gjyli, Goudjil, Igoa, Kahveci, Kalienko, Lorenzon, Macera, Manrique Fajardo, Nushi, Ouaatta, Parisi, Pasqualetto, Pesko, Pierri, Pinalli, Poppe, Santoro, Smirnova, Sorbara, Tensi & Tusha [IUCrData (2019). 4, x191222].
4-phenylphosphonato)zinc(II)], [Zn(C6H5O3P)], was obtained serendipitously by reacting zinc(II) acetate with a tetraphosphonate cavitand in DMF. The basic conditions of the reaction cleaved the phosphonate bridges at the upper rim of the cavitand, making them available for reaction with the zinc ions. The coordination polymer can be described as an inorganic layer in which zinc coordinates the oxygen atoms of the phosphonate groups in a distorted tetrahedral environment, while the phenyl groups, which are statistically disordered over two orientations, point up and down with respect to the layer. The layers interact through van der Waals interactions. Data analysis, structure solution, refinement and validation were conducted as part of a tutorial session during the Crystallographic Information Fiesta held in Naples, Italy, from 29 August to 3 September 2019, and organized by the Italian Crystallographic Association in partnership with the IUCr. See: Falvello, Lotti, Massera, Tarantino, Zema, Puschmann, Agbahoungbata, Andreo, Ashoka Sahadevan, Bismuto, Bonfant, Bonou, Carraro, De Zotti, di Biase, Fantini, Ferraboschi, Ferreira Custodio, Frigerio, Gallo, Gjyli, Goudjil, Igoa, Kahveci, Kalienko, Lorenzon, Macera, Manrique Fajardo, Nushi, Ouaatta, Parisi, Pasqualetto, Pesko, Pierri, Pinalli, Poppe, Santoro, Smirnova, Sorbara, Tensi & Tusha [IUCrData (2019). 4, x191222].

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 journal menu
journal menu










![[publCIF]](/logos/authorchecklist11.gif)