issue contents
July 2020 issue
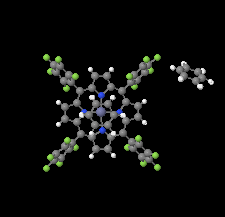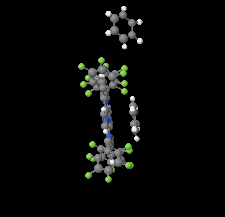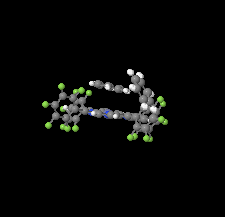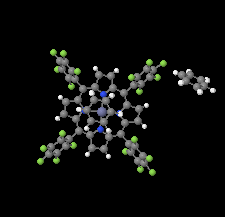
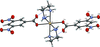
Cover illustration: The porphyrinato core of the zinc porphyrinato complex [5,10,15,20-tetrakis(pentafluorophenyl)porphyrinato]zinc(II) benzene disolvate is approximately planar, and the cation has no other ligating atoms than the four porphyrinato N atoms. The molecular complex exhibits point group symmetry ![]() with the central ZnII atom located on an inversion centre.
with the central ZnII atom located on an inversion centre. ![[pi]](/logos/entities/pi_rmgif.gif) -
-![[pi]](/logos/entities/pi_rmgif.gif) interactions between benzene solvent molecules and [Zn(TFPP)] units lead to multilayer packing structures. In addition, intermolecular C-H
interactions between benzene solvent molecules and [Zn(TFPP)] units lead to multilayer packing structures. In addition, intermolecular C-H F hydrogen bonding is observed between [Zn(TFPP)] molecules. See: Lin & Li [IUCrData (2020). 5, x200877].
F hydrogen bonding is observed between [Zn(TFPP)] molecules. See: Lin & Li [IUCrData (2020). 5, x200877].
inorganic compounds


 access
accessmetal-organic compounds


 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
accessorganic compounds


 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 journal menu
journal menu













![[publCIF]](/logos/authorchecklist11.gif)