issue contents
November 2018 issue
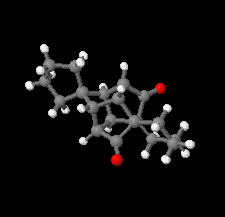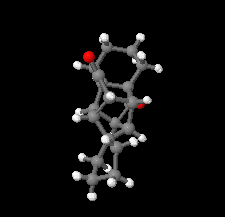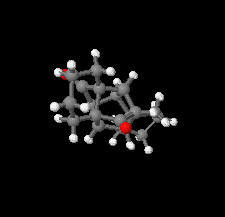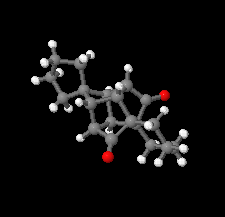
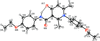
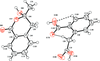
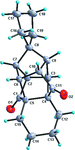
Cover illustration: D3-Trishomocubane and its derivatives are of interest for their use as targets for pharmaceutical applications and in medicinal chemistry as well as in asymmetric catalysis. Several amino functionalized D3-trishomocubane derivatives show significant biological activity and are NMDA receptor antagonists. In addition, some of them exhibit potent anti-TB activity, act as P2X7 receptor antagonists and exhibit anti-Parkinson's activity. The cage compound spiro[cyclopentane-1,11'-hexacyclo[7.6.0.01,6.06,13.08,12.010,14]pentadecane]-7',15'-dione was prepared from cheap and commercially available starting materials such as 1,4-hydroquinone in eight steps via Claisen rearrangement, Diels-Alder reaction and ring-closing metathesis (RCM) followed by acid-promoted rearrangement with Lewis acid. See: Kotha, Gunta, Cheekatla & Mhatre [IUCrData (2017). 3, x181590].
inorganic compounds


 access
accessmetal-organic compounds


 access
access

 access
access

 access
accessorganic compounds


 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 access
access

 journal menu
journal menu










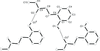
![[publCIF]](/logos/authorchecklist11.gif)